Interactions of Bacillus megaterium,
Pseudomonas putida and Pseudomonas brassicacearum;
the effects on plant growth (2013-2014)
Experiment
characteristics:
· Lycopersicon esculentum (cultivar “Tiny Tim”)
· Duration 128 days (17.04.2013–22.08.2013)
· Previously unused identical 2,5-litre polypropylene containers disinfected in 0,5% NaClO solution
· Soil mix containing 70% sieved commercial fertilized peat (particle size <3mm) & 30% sand (particle size 3-5mm) (v/w)
· Soil mix sterilized with γ-rays (30 kGy)
· 8 plants in each treatment block; 8 blocks in total (see below)
· 2 plants from each block in each growth chamber (randomized stratified block design)
· Daytime 23°C ~50% hum. (14h, light intensity >1000 μmol m-2 s-1), night 18°C ~50% hum.
· The identification of bacteria is based on 16S rRNA and biochemical characteristics
· Plants were inoculated immediately after sprouting; 2ml of each bacterial culture (grown for 24h in liquid LB broth at 30°C) was added
· Total crop was 1398 tomatoes (4,94kg), of which 944 ripened during the experiment (with an average weight of 4,5g)
Block design:
1) Control (abbr. C)
2) B.
megaterium (isolated from rhizosphere) (abbr. m)
3) P. putida (isolated
from rhizosphere) (abbr. p)
4) P. brassicacearum (isolated as an
endophyte) (abbr. b)
5) B.
megaterium & P. putida
6) B. megaterium & P. brassicacearum
7) P.
putida & P. brassicacearum
8) B.
megaterium & P. putida & P. brassicacearum
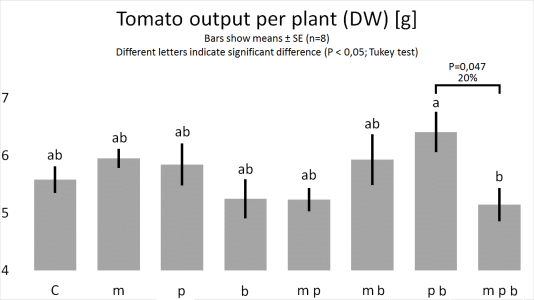
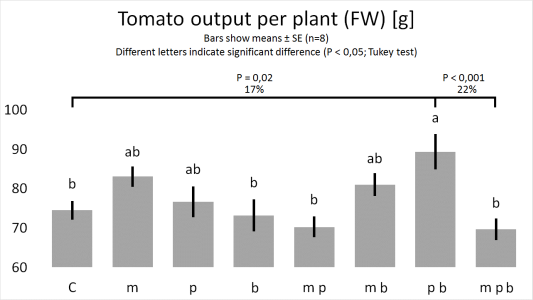
Figure
1. & 2.
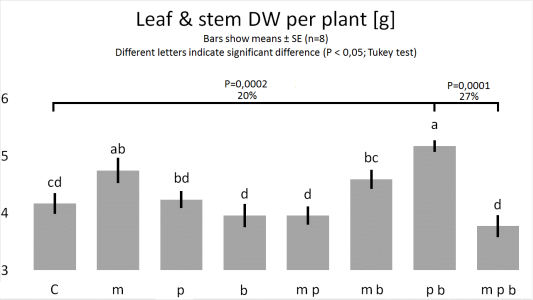
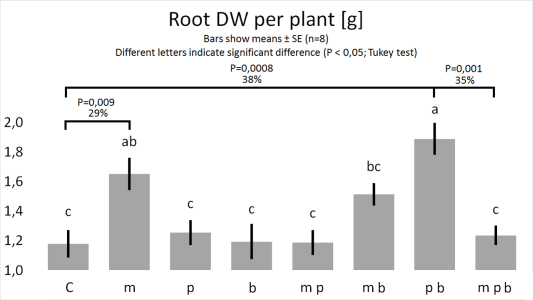
Figure
3. & 4.
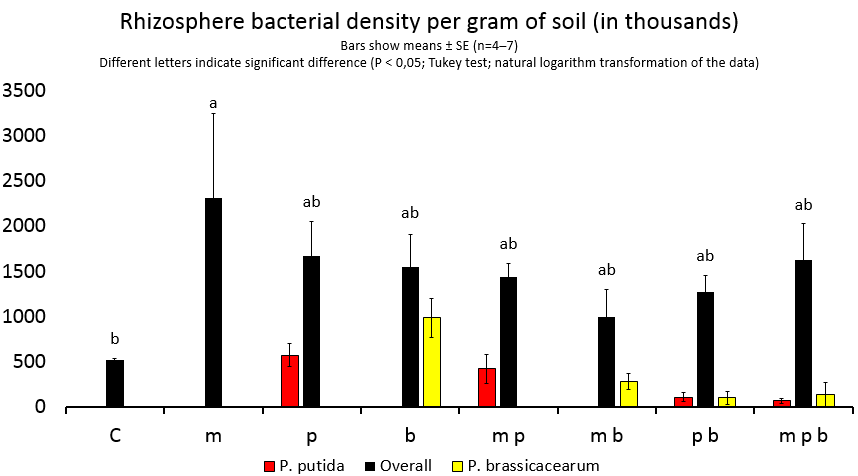
Figure 5.
Results
1. Bacillus megaterium improves the growth of plant roots.
2. Pseudomonas putida and Pseudomonas brassicacearum, combined, improve the growth of plants.
3. B. megaterium abolishes the joint plant growth promoting effects of P. putida and P. brassicacearum.
4. Ten out of 1398 tomatoes were deformed (“twin tomatoes”, Fig. 7.), eight of which originated from P. putida containing groups (P=0,04, Chi-square).
Brief look at the literature:
· P. brassicacearum produces N-Acyl homoserine lactones (AHL-s) with chain lenghts of 12–14 carbon atoms: 3OH-C12-AHL, 3OH-C14-AHL & C14-AHL. (Lalaouna et al. Appl. Environ. Microbiol. 2012)
· Pseudomonas sp. was found to produce an unusually long-chain 3O-C16-HSL. (Chang et al. PLOS ONE, 2012)
· Pseudomonas sp. which did not produce any quorum sensing (QS) molecules may play a role as a social cheater in the environment and may be a rider that exploits QS system. Social cheaters can derive the advantage and benefit of QS while avoiding the cost of producing the QS signalling molecules. (Chang et al. PLOS ONE, 2012)
· B. megaterium is capable of binding and oxidizing AHL-s with chain lengths of 12–16 carbons, irreversibly reducing their quorum activity. (Chowdhary et al. J Biotech, 2008)
My hypotheses:
· One of the Pseudomonas sp. (presumably P. brassicacearum) produces long-chain AHL-s that the other Pseudomonas sp. (presumably P. putida) is using (as a social cheat) for purposes from which the plant greatly benefits from.
· B. megaterium is oxidizing these long-chain AHL-s for its own energetic purposes, therefore disrupting quorum sensing between P. putida and P. brassicacearum, process known as quorum quenching.
I could not reproduce the results with
Arabidopsis thaliana in similar settings:

Figure 6. Above: copy of Fig. 3 for comparison. In the middle: First experiment with A. thaliana Col-0. Below: Second experiment with A. thaliana Col-0.

Figure
7. Upper two rows include photos of deformed tomatoes (n=8) from P. putida containing groups, the last
row includes photos of tomatoes (n=2) from control-group and B. megaterium + P. brassicacearum containing group.